Types of corrosion that affect rolling stock and how to reduce exposure | Sponsored | Railway Gazette International

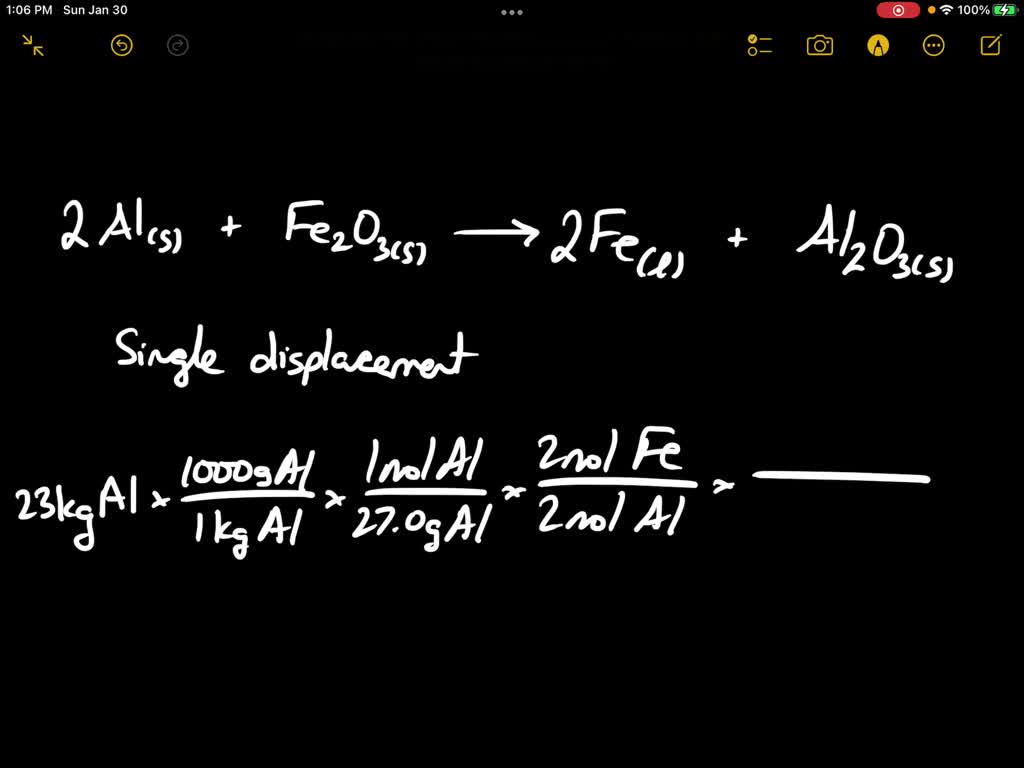
SOLVED: Write a balanced chemical equation based on the following description: the reaction of powdered aluminum and powdered iron(III) oxide produces solid aluminum oxide and liquid iron metal
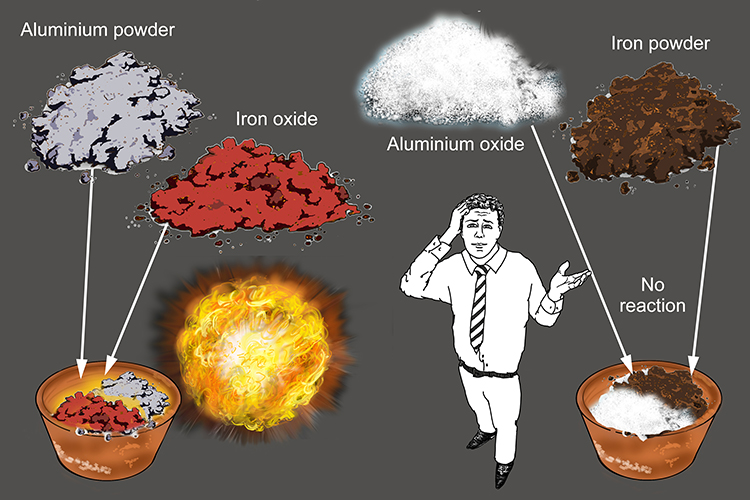
![Thermite By Jack Rjeili. What is Thermite? Mix of aluminum powder and iron oxide [1] Once ignited, produces extreme temperatures Produced from union. - ppt download Thermite By Jack Rjeili. What is Thermite? Mix of aluminum powder and iron oxide [1] Once ignited, produces extreme temperatures Produced from union. - ppt download](https://images.slideplayer.com/33/8235959/slides/slide_4.jpg)
Thermite By Jack Rjeili. What is Thermite? Mix of aluminum powder and iron oxide [1] Once ignited, produces extreme temperatures Produced from union. - ppt download

SOLVED:In the thermite reaction, iron(III) oxide is reduced by aluminum to give molten iron. Fe2 O3(s)+2 Al(s) →2 Fe(ℓ)+Al2 O3(s) If you begin with 10.0 g of Fe2 O3 and 20.0 g
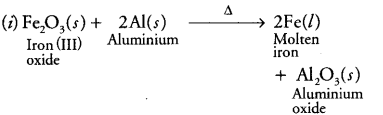
Write the balanced chemical equations for the following reactions and identify the type of reaction in each case. (i) In thermite reaction, iron (III) oxide reacts with aluminium and gives molten iron

An extensive study on the synthesis of iron based magnetic aluminium oxide nanocomposites by solution combustion method - ScienceDirect